Channels
Special Offers & Promotions
Avacta Life Sciences
Products
Contact Avacta Life Sciences
All articles from Avacta Life Sciences
Avacta announces results of initial evaluation of SARS-CoV-2 lateral flow antigen test with clinical samples
Feb 18, 2021
Avacta and Integumen plc collaborate for detection of SARS-COV-2 coronavirus in waste water
Jul 31, 2020
Excellent Results for Affimer Technology in Industry-Standard Immunogenicity Study Using Human Samples
Apr 6, 2017
Avacta and Glythera Announce Collaboration to Develop Novel, Potentially Highly Potent, Drug Class
Jul 16, 2016
Avacta Life Sciences Unveils Custom Seven Week Affimer Service to Revolutionise Life Sciences
Aug 11, 2015
Media Partners


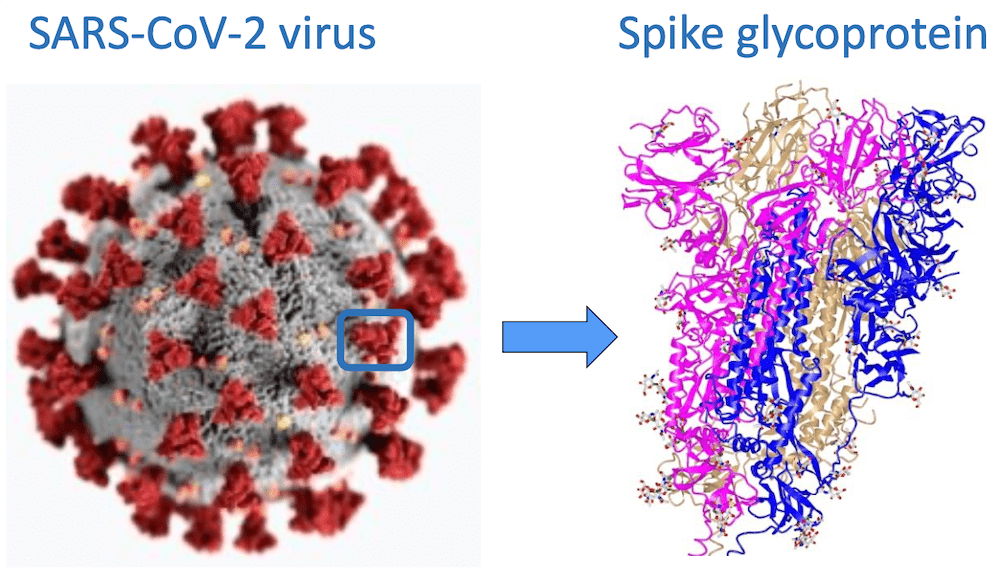 Avacta Group plc, the developer of innovative cancer therapies and diagnostics based on its proprietary Affimer® and pre|CISION™ platforms, is pleased to announce that the BAMS™ assay to detect the SARS-CoV-2 virus has been launched as a research kit by its partner Adeptrix (Beverly, MA, USA) and the assay has been presented by Bruker Scientific (Billerica, MA, USA), a leading mass spectrometer manufacturer, in a new application note...
Avacta Group plc, the developer of innovative cancer therapies and diagnostics based on its proprietary Affimer® and pre|CISION™ platforms, is pleased to announce that the BAMS™ assay to detect the SARS-CoV-2 virus has been launched as a research kit by its partner Adeptrix (Beverly, MA, USA) and the assay has been presented by Bruker Scientific (Billerica, MA, USA), a leading mass spectrometer manufacturer, in a new application note... Avacta Group plc, the developer of Affimer® biotherapeutics and reagents, is pleased to announce that it has begun work with the UK government’s CONDOR programme to evaluate and clinically validate the high throughput COVID-19 bead-assisted mass spectrometry (“BAMS™”) laboratory assay developed with Adeptrix. The COVID-19 National Diagnostic Research and Evaluation Platform (CONDOR), recently funded by the National Institute for Health Research...
Avacta Group plc, the developer of Affimer® biotherapeutics and reagents, is pleased to announce that it has begun work with the UK government’s CONDOR programme to evaluate and clinically validate the high throughput COVID-19 bead-assisted mass spectrometry (“BAMS™”) laboratory assay developed with Adeptrix. The COVID-19 National Diagnostic Research and Evaluation Platform (CONDOR), recently funded by the National Institute for Health Research... Avacta Group plc (AIM: AVCT), the developer of Affimer® biotherapeutics and reagents, is pleased to announce that it has entered into a collaboration with Cytiva, formerly known as GE Healthcare Life Sciences. The companies will develop and manufacture an Affimer-based point-of-care rapid test intended for screening of large populations to diagnose the COVID-19 coronavirus infection. The World Health Organisation recently....
Avacta Group plc (AIM: AVCT), the developer of Affimer® biotherapeutics and reagents, is pleased to announce that it has entered into a collaboration with Cytiva, formerly known as GE Healthcare Life Sciences. The companies will develop and manufacture an Affimer-based point-of-care rapid test intended for screening of large populations to diagnose the COVID-19 coronavirus infection. The World Health Organisation recently....
 Avacta Group plc, the developer of Affimer® biotherapeutics and research reagents, has announced it has appointed Professor Gerard Evan, FRS, to its therapeutics Scientific Advisory Board. Professor Evan brings world-leading expertise in immuno-oncology and cancer stem cell research...
Avacta Group plc, the developer of Affimer® biotherapeutics and research reagents, has announced it has appointed Professor Gerard Evan, FRS, to its therapeutics Scientific Advisory Board. Professor Evan brings world-leading expertise in immuno-oncology and cancer stem cell research...
