Channels
Special Offers & Promotions
Spinning Disk Super Resolution Imaging uses Piezo Control to Enhance Stability
Super resolution microscopy has the ability to produce biological images that show molecular level structures.
 Although the technique is limited when it comes to looking at live cells a number of research groups from around the world are working on overcoming these limitations, including a team led by Dr Ann Wheeler, Head of Imaging at the Queen Mary University of London’s Blizard Institute*, and Professor Martin Knight of Queen Mary’s School of Engineering and Materials Science.
Although the technique is limited when it comes to looking at live cells a number of research groups from around the world are working on overcoming these limitations, including a team led by Dr Ann Wheeler, Head of Imaging at the Queen Mary University of London’s Blizard Institute*, and Professor Martin Knight of Queen Mary’s School of Engineering and Materials Science.
When Dr Wheeler, Prof Knight and their team needed improved stability for their new spinning disk super resolution imaging (SDSI) microscope, they turned to precision positioning specialists, PI Ltd for help. The solution based on PI’s piezo driven, capacitive sensor feedback nano-positioning technology helped to provide the long-term stability required.
The new SDSI microscope was developed by combining the single focus plane technology of a spinning disk system with the super resolution techniques of Photoactivation Light-Microscopy (PALM) and Stochastic Optical Reconstruction Microscopy (STORM).
A confocal or spinning disk microscope, which is typically used to generate images of live cells, has a spatial resolution limited to about 200nm. The technique uses a low laser power to rapidly scan live cells collecting a stack of image planes as it steps through in the focus direction. The images are then recombined to produce a 3D fluorescent picture of the sample.
Currently PALM and STORM super-resolution techniques use TIRF to collect fluorescent images over an extended time period and then use complex algorithms to process the data. The processed images can resolve artefacts with a resolution of down to 30nm. A good signal to noise ratio is needed to ensure the algorithms are effective and this is achieved by using a high laser power for TIRF illumination. The power of the laser can cause photo-bleaching in live cells so limiting the technique’s range of use. Also the penetration depth of TIRF is limited to 100nm so preventing access to the cell nucleus.
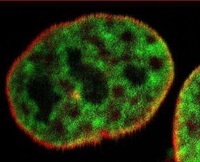
The microscope at the Blizard uses a spinning disk to acquire the image and then processes the data using super resolution algorithms. Acquiring the images using the spinning disc allows both a lower laser power to be used and penetration depths to reach the level of the nucleus. The technique has opened up new opportunities in biological imaging, producing pictures with a resolution of 80nm and allowing artefacts, such as protein complexes in the nucleus to be viewed.
Each set of data taken from a single image plane is acquired over several minutes. During data collection the sample must remain stable relative to the focal plane. Any drift in the microscope due to thermal effects must be compensated for. Despite leaving the microscope to stabilise over several hours and keeping it in a temperature controlled environment the Blizard were seeing drift in the focal plane.
Following a visit by PI and discussions about the problem the company recommended that the Blizard use the P-725 PIFOC piezo driven nanopositioning objective drive. The PIFOC incorporates PI’s own capacitive position sensor to provide position control and stability to the nanometre scale over a wide temperature range. The capacitive sensor measures the air gap between two plates of the same material placed only a few tens of microns apart. The design minimises the effects of temperature change ensuring nanometre precision is maintained over a wide operating range. Once fitted to the microscope the PIFOC was successfully used to compensate for drift during the measurement cycle. Implementation of the PIFOC has provided the Blizard with the stability required in the SDSI to generate an extensive range of data. The data has shown the viability of SDSI as an enhancement to existing techniques through the ability to collect super resolution images with good signal to noise in any selected axial plane within a cell. PI would like to thank Dr Ann Wheeler of The Blizard Institute, QMUL for her help in writing this article.
For more information please see the following research article: Super-Resolution Imaging Strategies for Cell Biologists Using a Spinning Disk Microscope Naveen A Hosny, Mingying Song, John T Connelly, Simon Ameer-Beg, Martin M Knight, Ann P Wheeler
Media Partners


