Channels
Special Offers & Promotions
Indica Labs Receives First FDA Clearance for HALO AP Dx Digital Pathology Platform
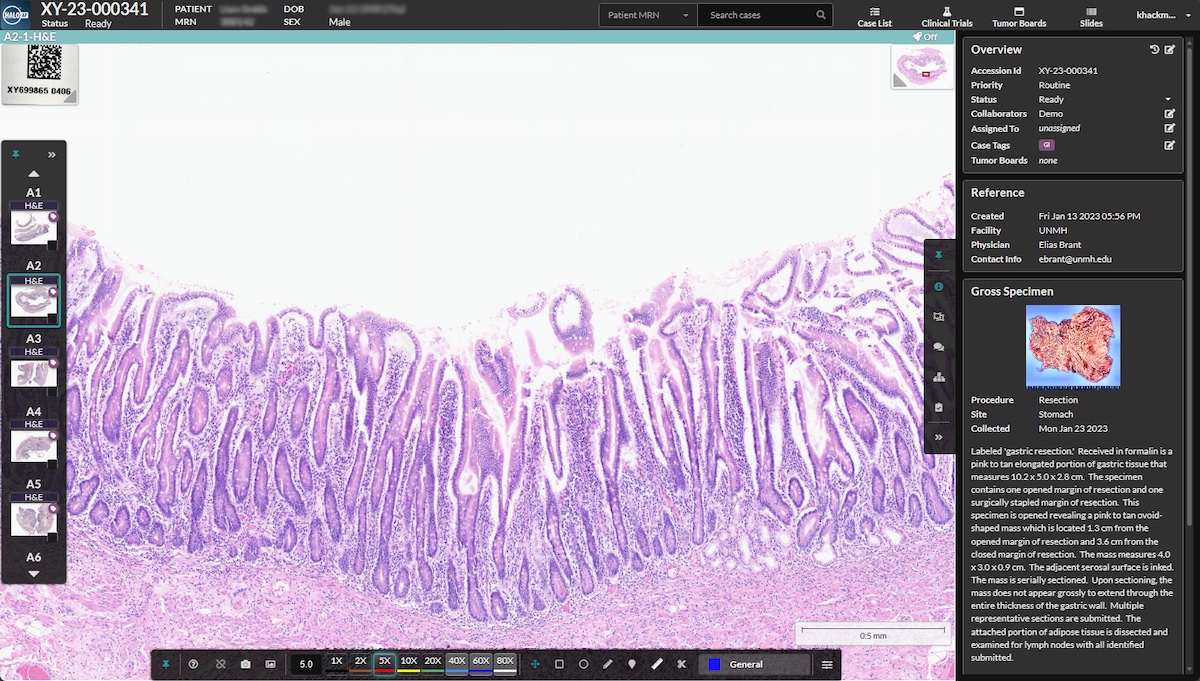
Indica Labs, the leading provider of digital pathology solutions, announce that it received FDA clearance for HALO AP Dx, an enterprise digital pathology platform, for primary diagnosis. This 510(k) clearance allows HALO AP Dx to be utilized in conjunction with the Hamamatsu NanoZoomer® S360MD Slide scanner for in-vitro diagnostic use and sets the stage for use with additional FDA-cleared slide scanners in the future.
HALO AP® is an enterprise digital pathology platform that is deployed worldwide and has transformed the way pathologists and researchers analyze, manage, and utilize digital pathology images. To address the complex needs of modern pathology laboratories, Indica Labs created HALO AP® and HALO AP Dx with a comprehensive suite of diagnostic tools and features for efficient and accurate image evaluation, management, and collaboration. In the US, Indica Labs will continue to offer HALO AP® for RUO applications and will now offer HALO AP Dx for primary diagnosis.
"Our team is thrilled to achieve this FDA clearance for HALO AP Dx," said Steven Hashagen, founder and CEO of Indica Labs. "Indica Labs is dedicated to improving patient care by providing an ideal environment for the practice of pathology that supports both current and future technologies, ensuring pathologists have the best tools available at their fingertips and are prepared for whatever the future of digital pathology holds."
Owing to the company’s commitment to open pathology, the platform is highly interoperable. HALO AP Dx users can adapt the platform to meet their needs and incorporate it into their existing laboratory workflows with ease, creating a custom-built environment for digital pathology and for a variety of pathologic specialties, even those with unique technical requirements. HALO AP Dx is flexible and can be deployed in either a cloud-based or on-premises environment, granting users freedom of choice as to how their data is stored.
In addition to streamlining the diagnostic process, HALO AP Dx enables both remote work and remote consultations, features that are particularly crucial as case numbers increase amid a worsening pathologist shortage. With slides available for review from any location the moment they are scanned, pathology labs can run an uninterrupted pathology service with round-the-clock, global collaboration without having to ship a single slide.
Hashagen added, "This 510(k) clearance for HALO AP Dx enables pathologists in the US to deliver timely and accurate diagnoses, expediting the development of treatment plans and ultimately resulting in patients being treated sooner, which enhances patient care. We’ve already witnessed these improvements to patient care in the UK, where the platform is CE-IVDR marked for in-vitro diagnostic use, and we look forward to similar outcomes here in the US."
HALO AP Dx is a complete platform for pathology and a centralized solution for primary diagnosis, collaboration, tumor boards, and second opinions. Its powerful integration engine brings together diverse software and hardware functionalities. With HALO AP Dx, pathology practices gain not only a robust diagnostic workspace but also a collaborative hub that fosters innovation, adaptability, and future AI readiness. Leading hospitals, CROs, clinical laboratories, government and academic organizations trust HALO AP® to provide the power and usability needed to practice modern pathology.
To learn more about HALO AP Dx and to see a live demo, register for Indica Labs’ upcoming webinar. "Introducing HALO AP Dx: An Enterprise Pathology Platform for Primary Diagnosis" which will take place on Thursday, June 13 at 11AM EDT.
HALO AP Dx (K232833) is FDA-cleared for primary diagnostic use with the Hamamatsu NanoZoomer® S360MD Slide scanner in the US. In addition, HALO AP Dx provides built-in compliance and certifications with FDA 21 CFR Part 11, ISO 13485:2016, and HIPAA.
HALO AP® is CE-IVDR marked for in-vitro diagnostic use in Europe, the UK, and Switzerland. HALO AP® is For Research Use Only in the US and is not FDA cleared for clinical diagnostic use. In addition, HALO AP® provides built-in compliance and certifications with FDA 21 CFR Part 11, ISO 13485:2016, HIPAA, and GDPR.
About Indica Labs
Indica Labs is the global leader in AI-powered digital pathology software and services. Our flagship HALO® and HALO AI platform revolutionizes quantitative evaluation of whole slide images. HALO Link provides collaborative image management while HALO AP® and HALO AP Dx deliver enterprise digital pathology for primary diagnosis with regulatory clearances in multiple markets. Through a commitment to open pathology, performance, scalability, and ease-of-use, we help pharma companies, diagnostic labs, hospitals, research organizations, and Indica’s own Cloud and Pharma Services make discoveries and diagnoses that transform patient care and scientific discovery.
Media Partners


